Which of the Following Has No Net Dipole Moment
The size of a dipole is. Choose the statement that best describes the PbCl4 molecule in the gas.

Solved 1 Which Of The Following Has No Net Dipole Moment O Chegg Com
In understanding molecular polarity you need to take the whole structure into account.

. O CHCI O NF3 N20 TeO HSe check_circle Expert Answer. Based on symmetry alone we know that H 2S is the only one of these molecules that has a dipole moment. Which of the following molecules has no dipole moment.
Which of the following has a net dipole moment. Which of the following does not have a molecular dipole moment. C The electrons in a polar bond are found nearer to the more electronegative element.
In contrast the H 2 O molecule is not linear part b in Figure 228. A BF3 B NCl3 C H2Se D CH3Cl. Want to see the step-by-step answer.
Which of the following has no net dipole moment. Which of the below molecules has no net dipole moment. 101Which of the following has no net dipole momentN2O NF3 H2Se TeO3 CH3Cl.
B Dipole moments result from the unequal distribution of electrons in a molecule. A molecule which has a symmetrical geometry will. As a result the CO 2 molecule has no net dipole moment even though it has a substantial separation of charge.
Carbon tetrachloride has no net dipole moment because of. 2 CCIA 3 BeF24 SO Open in App. Between all these molecules NH3 will have net dipole moment as it is pyramidal in shape with 1 lone pair of e- on the top of the pyramidand 3-H atoms going downThe bond.
We can see that in one plane two chlorine and a bromine atom are there and hence they will not be able to cancel out each others effect and as a result this molecule will also have a net. Your reasoning is correct as far as the parts of the molecule are concerned. Which of the following has no net dipole moment.
Answered Aug 28 2021 by MehulKumar 187k points selected Aug 30 2021 by Ritwik. In the case of Cl2 the 2 atoms are identical so no polarization of. 95 130 ratings play-rounded-fill.
C H C l 3 has net dipole moment as the bond dipoles do not cancel each other. Each CO bond in CO 2 is polar yet experiments show that the CO 2 molecule has no dipole moment. A SCl2 B H2O C CF4 D BrCl.
H 2 O d. AskedJun 5 2019in Chemistryby Arnika Singh736kpoints class-11. Check out a sample QA.
Among all the given options TeO 3 has no net dipole moment because the central atom has three bonded atoms one bond being a double bond and has no lone pairs and the geometry is. When two electrical charges of opposite sign and equal magnitude are separated by a distance an electric dipole is established. Whereas other molecules such as C H 4 C O 2 and C C l 4 have zero dipole moment as the bond dipoles completely cancel each other.
Because the two CO bond dipoles in CO 2 are equal in magnitude and.
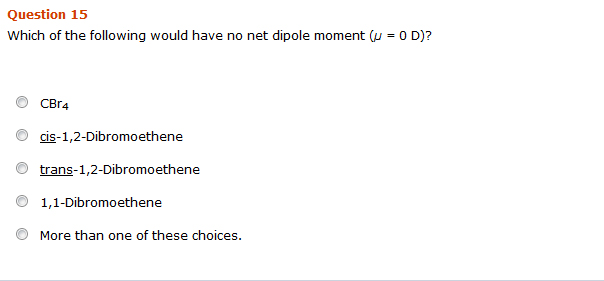
Solved Which Of The Following Would Have No Net Dipole Chegg Com

Solved Which Of The Following Has No Net Dipole Moment A Chegg Com

Which Of The Following Has No Net Dipole Moment

Which Of The Following Alkanes Has No Net Dipole Moment Youtube
Comments
Post a Comment